Description
Axis-Shield Homocysteine EIA microtitre plate ELISA (96 Well)
Product Code: FHCY100
Axis-Shield Homocysteine EIA DataSheet
Assay Principle
Axis-Shield Homocysteine Enzyme Immunoassay (EIA) is an enzyme immunoassay for the determination of homocysteine (Hcy) in serum or plasma.
Protein-bound Hcy is reduced to free Hcy and enzymatically converted to S-adenosyl-L-homocysteine (SAH) in a separate procedure prior to the immunoassay. The following solid-phase ELISA is based on competition between SAH in the sample and immobilised SAH bound to the walls of the microtitre plate for binding sites on a monoclonal anti-SAH antibody. After a wash stage to remove unbound antibody, a secondary HRP-labelled rabbit anti-mouse antibody conjugate is added. The peroxidase activity is measured after addition of substrate and the absorbance is inversely related to the concentration of HCY in the sample.
Measuring Range
The calibrator range is from 2 to 50 µmol/L.
Assay Precision
Precision of the Axis-Shield Homocysteine EIA assay was evaluated according to NCCLS Protocol EP5. Three levels of controls were assayed for 20 days with 4 replicates per run at each level. Precision data are summarised in Table 1.
Table 1: Precision
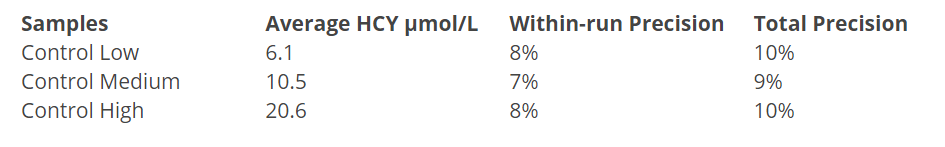
Limit of Quantification
The quantification limit (CV < 20%) is 1.0 µmol/L
Linearity of diluted plasma samples
If the homocysteine concentration of a sample exceeds the range of the calibration curve, the sample should be diluted with Reagent A and reanalysed.
The linearity was evaluated by diluting four high patient samples with varying amounts of Reagent A as diluent.
Linear regression analysis gave:
Slope: 0.98
Intercept: -0.4µmol/L
Correlation coefficient: r2: 0.99
Method Comparison
The Axis-Shield Homocysteine EIA was compared to the University of Bergen HPLC method. A comparison of 164 patient samples ranging from 3-37µmol/L homocysteine gave the linear regression shown below.
Slope: 0.94
Intercept: -0.09 µmol/L
Correlation coefficient: (r2): 0.94
Axis-Shield Homocysteine EIA (96 well) – FHCY100
Axis-Shield Homocysteine Control kit (tri-level, 1.5mL each) – FHCY200
1 Review
-
its good
The company is known for its good quality products that satisfy a customer

![HE4 EIA [CE] HE4 EIA [CE]](https://cdn11.bigcommerce.com/s-ryt90hjx0j/images/stencil/590x590/products/38361/55331/logo-diasource__09422.1773239006.png?c=1)


