T-Cell Grade Mouse Type II Collagen, 0.5 mg, lyophilized
- SKU:
- 445-2026
- Size:
- 0.5 mg
- Shipping:
- Gel Packs
- Storage:
- 4C, in the dark
Description
T-Cell Grade Mouse Type II Collagen, 0.5 mg, lyophilized - Cat Number: 2026 From Chondrex.
Research Field: Arthritis, ECM
Clonality: N/A
Cross-Reactivity:
Host Origin: N/A
Applications: N/A
Isotype: N/A
Detection Range: N/A
Sample Type: N/A
Concentration: N/A
Immunogen:
DESCRIPTION: Highly purified heat-denatured type II collagen
APPLICATION: Use as a stimulating antigen for T-cells from experimental animals and humans in in vitro culture
systems. For example, use in T-cell proliferation assays and in establishing type II collagen
specific T-cell lines. Type I, II, III, and even Type IV collagen share highly conserved amino acid
sequences but have unique immunological features. Therefore, it is useful to use different types
of collagen as a control for studying the epitope specificity of T-cells in collagen-induced arthritis
(CIA) and other disease models.
QUANTITY: 0.5 mg
FORM: Lyophilized powder
SOURCE: Mouse sternal cartilage
MOLECULAR WEIGHT: 300 kDa
PURITY: Purified by CM-cellulose chromatography
>99% by SDS-PAGE, free of pepsin and proteoglycans
USAGE: Soluble in acidic and neutral buffers, and in cell culture media. In order to use this product to
assay T-cell proliferation responses, Chondrex, Inc. recommends dissolving this product in
0.05M phosphate buffered saline rather than cell culture media at 1 mg/ml before use.
STORAGE: 4°C in the dark, as supplied. Denatured collagen dissolved in acidic solution is also stable at -
20°C for one year but will gradually degrade under neutral conditions.
STABILITY: 2 years
NOTES: N/A
REFERENCES: R. Trelstad. et al. Biochemistry 9: 4993-4998 (1970)
E. Miller. Biochemistry 10: 1652-1659 (1971)
K. Terato. et al. Brit. J. Rheum. 35: 828-838 (1996)
INTRODUCTION
T-cells play a crucial role in inducing collagen-induced arthritis (CIA) as type II collagen is a T-cell dependent antigen. However, it is difficult to study T-cell responses to type II collagen even in CIA high responder animals by T-cell proliferation assay using spleen and lymph node cells due to several reasons: 1) Collagen is not a strong T-cell antigen and T-cell populations specific to collagen are low. 2) T-cell proliferation responses will be masked due to the high background levels caused by polyclonally stimulated B-cells with adjuvant.
B-cells, even from animals immunized with incomplete adjuvant, proliferate spontaneously. 3) Macrophages activated by adjuvant components might release a large amount of immuno-suppressive cytokines, which inhibit T-cell proliferation. 4) Pepsin, which is commonly used for solubilizing collagen and a common contaminant in collagen preparations, is a strong T-cell antigen, and creates significantly high false positive results.
In order to determine true T-cell responses to collagen, it is important to use a T-cell enriched fraction from spleen or lymph node cells, and to use highly purified, pepsin free collagen preparations as a T-cell stimulation antigen in vivo. Type II collagen is used as an immunizing antigen for inducing arthritis particularly in rodents and monkeys. However, depending upon the research purpose, animals may be immunized with type I collagen or receive collagen implants.
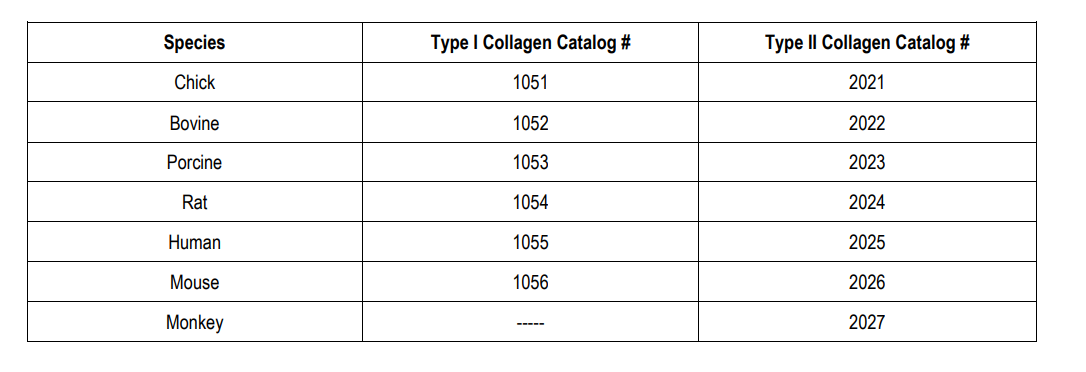
Chondrex, Inc. provides various species of highly purified, heat-denatured T-cell grade type I and type II collagen for various purposes and immunological studies on collagen and collagen related materials (i.e. T-cell proliferation assays and establishing T-cell lines and T-cell hybridoma cells. In addition, for T-cell epitope studies, CNBr-digested type II collagen fragments can be provided upon request. Type I collagen can be used as a control antigen for T-cell epitope studies in the CIA model and other autoimmune models as type I and type II collagen share more than 80% of their amino acid sequences, even if their immunological features differ.
PROCEDURE FOR IN VITRO T-CELL PROLIFERATION ASSAY
Immunization
Immunize animals such as mice, rats or monkeys with type II collagen emulsified with CFA (Catalog # 7001) or IFA (Catalog # 7002). Follow the immunization protocol for inducing collagen-induced arthritis in individual animals. Collect spleen or lymph node cells on day 8-12 after immunization. In the case of monkeys, use peripheral blood mononuclear cells (PBMC) or an underarm lymph node.
NOTE 1: It is not necessary to use the immunization protocol for inducing CIA in these animals, but it might be convenient to follow the protocol. For example, immunize mice with 100 µg of type II collagen emulsified with CFA, rats with 200 µg of type II collagen emulsified inIFA, and monkeys with 250 µg of type II collagen emulsified with CFA.
NOTE 2: T-cell proliferation responses and their epitope specificity are not dependent on the conformational structure of type II collagen or the type of adjuvant. Identical T-cell responses are observed in mice immunized with either native or denatured collagen emulsified with both CFA and IFA. Therefore, for T-cell studies, denatured collagen and even CB peptide fragments can be used as immunizing antigens. On the other hand, antibody specificity and induction of arthritis are strongly dependent on the conformational structure of type II collagen and the type of adjuvant (see “Successful Induction of Arthritis in Mice” and “Successful Induction of Arthritis in Rats”).
Purifying T-Cells
1. Pack 0.6 g of pre-washed nylon wool into a 10 ml syringe, soak with PBS, and autoclave for 20 minutes. Replace the PBS with Dulbecco’s Minimum Essential Medium (DMEM) supplemented with 1% homologous normal serum, penicillin-streptomycin (1X), and
2-mercaptoethanol (2-ME - 5 x 10-5M). This is called complete DMEM.
NOTE: Do not use fetal calf or bovine serum as T-cells spontaneously react to heterologous serum components. In the case of monkeys, use Protein A-adsorbed autologous serum or plasma (Protein A removes immunoglobulins including antibodies against collagen).
2. Prepare a single cell suspension from spleen or lymph node cells from mice immunized with type II collagen (or type I collagen), wash 3 times with DMEM, and then suspend the cells in 1-2 ml of complete DMEM (approximately 1 x 108 cells/ml).
3. Load the cell suspension onto the column. Incubate the column for 30 minutes in a CO2 incubator at 37°C. Elute unbound cells with 20 ml DMEM and then centrifuge at 800 rpm for 8 minutes. Suspend the enriched T-cells at 4 8 x 106 cells/ml in complete DMEM.
Preparing Antigen Presentation Cells (APC)
1. Use spleen or thymus cells obtained from young, normal congenic animals (6-8 weeks old) as APC. Wash the cells with DMEM 3 times
and suspend at 1 x 107 cells/ml in complete DMEM.
NOTE: In the case of monkeys, use Mytomycin C (an inhibitor of DNA synthesis) treated PBMC isolated by the Ficoll-Hypaque method as APC.
2. Irradiate the cells with 2000 rad = 20 Gy.
NOTE: Do not disturb the cells after irradiation and treatment with Mytomycin C (50 µg/ml for 1 hour).
T-Cell Proliferation Assay
1. Dissolve T-cell grade type II collagen (or type I collagen) at 1 mg/ml with warm PBS or DMEM and then centrifuge at 10,000 rpm for 3 minutes at room temperature. Transfer the supernatant to a sterile tube and store at -20°C until needed.
NOTE: T-cell grade type II collagen (or type I collagen) has been denatured by heating. Type II collagen (or type I collagen) used for Tcell stimulation should be denatured as APC are not capable of processing native type II collagen (or type I collagen) in such a short culture period.
2. Add 0.1 ml of enriched T-cells (4-8 x 106 cells/ml), 0.1 ml of APC (1 x 107 cells/ml), and 10-20 µl of 1 mg/ml T-cell grade type II collagen (or type I collagen) into the wells of a 96-well flat bottom plate. Culture the cells in a CO2 incubator at 37°C with 5% CO2 for 3 days.
NOTE 1: In the case of monkeys, optimizing the T-cells/APC ratio (in general, an equal number of T-cells and APCs is recommended) is critical to obtain the maximum T-cell responses to collagen.
NOTE 2: Do not use round bottom plates.
3. Add 3H-labeled thymidine (0.5-1.0 mCi/well) on day 3 and culture for an additional 15-18 hours. Harvest the cells on a glass filter and assay for the incorporated 3H by scintillation counting.






