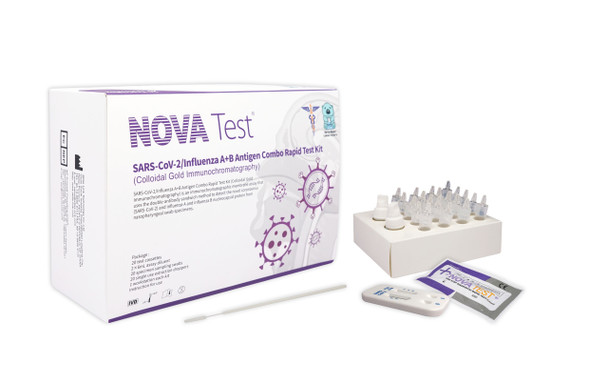
NOVATest CoV/Flu Antigen Combo Rapid Test Kit
- SKU:
- nCovf-501
- Availability:
- Next week
- Minimum Purchase:
- 100 units
- Size:
- 20T
Description
NOVATest CoV/Flu Antigen Combo Rapid Test Kit | nCovf-501 | NOVA Test
INTENDED USE:
This kit is used for in vitro rapid qualitative detection of novel coronavirus (SARS-CoV-2) and influenza A+B antigens in nasopharyngeal swab samples. The product is designed to be used for population screening early phase for disease outbreaks. Testing is limited to laboratories certified. Results are for the identification of SARS-CoV-2 nucleocapsid and influenza A and influenza B antigen. This antigen is generally detectable in upper
respiratory samples during the acute phase of infection.
NOVA Test®SARS-CoV-2/Influenza A+B Antigen Combo Rapid Test Kit provides preliminary test results, with negative results don’t preclude SARS-CoV-2 or influenza infection. Can not be used as the sole basis for treatment or other management decision. For in vitro diagnostic use only.
Priniple:
The NOVA Test®SARS-CoV-2/Influenza A+B Antigen Combo Rapid Test Kit (Nasopharyngeal Swab) is a qualitative membrane-based immunoassay for the detection of SARS-CoV-2 Antigens in human nasopharyngeal swab specimen. SARS-CoV-2 antibody is coated in test line region.
During testing, the specimen reacts with SARS-CoV-2 antibody-coated particles in the test. The mixture then migrates upward on the membrane by capillary action and reacts with the SARS-CoV-2 antibody in test line region. If the specimen contains
SARS-CoV-2 Antigens, a colored line will appear in test line region as a result of this. If the specimen does not contain antigens to SARS-CoV-2, no colored line will appear in
the test line region, indicating a negative result. To serve as a procedural control, a colored line will always appear in the control line region, indicating that the proper volume of specimen has been added and membrane wicking has occurred.
The Influenza A+B Rapid Test (Nasopharyngeal Swab) is a qualitative, lateral flow immunoassay for the detection of Influenza A and Influenza B antigen in human nasopharyngeal swab specimen. In this test, antibody specific to the Influenza A and Influenza B is separately coated on the test line regions of the test. During testing, the extracted specimen reacts with the antibody to Influenza A and/or Influenza B that are
coated onto particles. The mixture migrates up the membrane to react with the antibody to Influenza A and/or Influenza B on the membrane and generate one or two colored lines in the test regions. The presence of this colored line in either or both of the test regions indicates a positive result. To serve as a procedural control, a colored line will always appear in the control region if the test has performed properly.
Reagents:
The test contains anti-SARS-COV-2, anti-Influenza A and anti-Influenza B as the capture reagent, anti-SARS-COV-2, anti-Influenza A and anti-Influenza B as the detection reagent.
Storage Conditions:
The swab specimen in dry and sterile condition is stable for up to 8 hours at room temperature and 24 hours at 2-8°C.
Data Sheet:
1 Review
-
good support
my experinec with Gentaur is really good. I am in love with their products and services.


![SARS-CoV-2/Influenza A+B antigen Combo Rapid Test Cassette [CE] SARS-CoV-2/Influenza A+B antigen Combo Rapid Test Cassette [CE]](https://cdn11.bigcommerce.com/s-ryt90hjx0j/images/stencil/590x590/products/38732/55634/logo-diasource__24968.1773239012.png?c=1)

