Description
MMQCI QIAstat-Dx RP SARS-CoV-2 Control
Panel Part Number: M360v1.1
Kit Contains: 12 tubes x 300µL
4 each Positive A, Positive B1, and Negative
QIAstat-Dx RP SARS-CoV-2 Control Panel DataSheet
INTENDED USE:
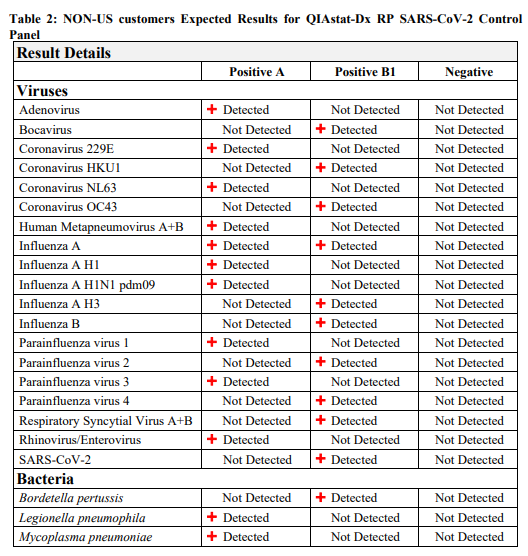
QIAstat-Dx RP SARS-CoV-2 Control Panel is intended for use as an external positive and negative quality control to monitor the performance of in vitro laboratory nucleic acid testing procedures for the qualitative detection of bacterial and viral pathogens: Adenovirus, Bocavirus, Coronavirus 229E, Coronavirus HKU1, Coronavirus NL63, Coronavirus OC43, Severe Acute Respiratory- Coronavirus 2 (SARS-CoV-2), Human Metapneumovirus A+B, Influenza A, Influenza A H1, Influenza A H1N1 pdm09, Influenza A H3, Influenza B, Parainfluenza virus 1, Parainfluenza virus 2, Parainfluenza virus 3, Parainfluenza virus 4, Respiratory Syncytial Virus A+B, Rhinovirus/Enterovirus, Bordetella pertussis, Chlamydophila pneumoniae, Legionella pneumophila and Mycoplasma pneumoniae on the QIAstat-Dx® Respiratory SARS-CoV-2 Panel performed on QIAstat-Dx systems. QIAstat-Dx RP SARS-CoV-2 Control Panel is composed of synthetic RNA specifically designed for and intended to be used solely with the QIAstat-Dx Respiratory SARS-CoV-2 Panel.
PRODUCT SUMMARY and PRINCIPLE:
QIAstat-Dx RP SARS-CoV-2 Control Panel is composed of 3 controls, QIAstat-Dx RP Positive A, QIAstat-Dx RP Positive B1, and QIAstat-Dx RP Negative. QIAstat-Dx RP Positive A and QIAstat- Dx RP Positive B1 contain surrogate control material composed of synthetic nucleic acid corresponding to genome segments of pathogens listed in Table 1 and 2. QIAstat-Dx RP Negative contains no nucleic acid.
Routine use of quality controls that are consistent lot to lot assists the laboratory in identifying shifts, trends, and increased frequency of random errors caused by variations in the test system, such as failing reagents. Early investigation can prevent failed assay runs.
COMPOSITION:
The QIAstat-Dx RP SARS-CoV-2 Control Panel is comprised of 12 tubes, 4 tubes of QIAstat-Dx RP Positive A, 4 tubes of QIAstat-Dx RP Positive B1, and 4 tubes of QIAstat-Dx RP Negative, 300µL each. QIAstat-Dx RP Positive A and QIAstat-Dx RP Positive B1 contain synthetic RNA suspended in a non-infectious solution of buffers, preservatives and stabilizers. QIAstat-Dx RP Negative contains buffers and preservatives. Table 1 and 2 lists the pathogens that are monitored by the QIAstat-Dx RP SARS-CoV-2 Control Panel when tested by the QIAstat-Dx Respiratory SARS-CoV-2 Panel on the QIAstat-Dx systems.
INSTRUCTIONS FOR USE:
- Allow the control to be tested to come completely to room temperature (18° – 25°C).
- Use the control as provided. DO NOT DILUTE.
- Prepare a QIAstat-Dx Respiratory SARS-CoV-2 Panel Cartridge according to the QIAstat- Dx Respiratory SARS-CoV-2 Panel Instructions for
- Open the sample lid of the main port on the front of the QIAstat-Dx Respiratory Panel Cartridge according to the QIAstat-Dx Respiratory SARS-CoV-2 Panel Instructions for
- Immediately before use, mix the control thoroughly by inverting 3-5 times and tap the tube several times on the bench to remove any control caught in the cap before opening the
- Use the supplied transfer pipette to draw up the liquid control to the third fill line on the pipette (300µL).
- Carefully transfer 300µL of sample volume into the main port on the QIAstat-Dx Respiratory SARS-CoV-2 Panel Firmly close the sample lid of the main port until it clicks.
- Visually confirm that the sample has been loaded by checking the sample inspection window in the cartridge.
- The controls are single use Discard control tube after use according to your local and federal regulations.
- Continue to process the control according to QIAstat-Dx Respiratory SARS-CoV-2 Panel Instructions for
PRECAUTIONS, WARNINGS and LIMITATIONS:
- Do not Use the control as provided.
- This product is intended for in vitro analytical
- This product is only for use with QIAstat-Dx Respiratory SARS-CoV-2 Panel on the QIAstat- Dx It does not contain the entire genome of the respiratory pathogens listed in Table 1 and 2.
- This product is not intended for use as a substitute for the internal controls provided in the QIAstat-Dx Respiratory SARS-CoV-2
- Appearance: Positive A & B1 controls are slightly cloudy and Negative control is
- This product does not contain any biological material of human or animal Universal Precautions are NOT required when handling this product.
- Quality control materials should be used in accordance with local, state, federal regulations and accreditation
- QIAstat-Dx RP SARS-CoV-2 Control Panel cannot be cloned, sold, or transferred without the explicit written consent of MMQCI.
STORAGE and STABILITY:
QIAstat-Dx RP SARS-CoV-2 Control Panel should be stored frozen (-25°C to -15°C). Unopened QIAstat-Dx RP SARS-CoV-2 Control Panel material is stable through the expiration date printed on the kit label when stored frozen. QIAstat-Dx RP Positive A, QIAstat-Dx RP Positive B1, and QIAstat-Dx RP Negative are for single use only. Discard after use according to your local and federal regulations.
EXPECTED VALUES:
Table 1 and Table 2 list the expected results when the controls are analyzed by QIAstat-Dx Respiratory SARS-CoV-2 Panel. Note that the reported Ct/EP may vary run to run, and among different instruments and laboratories.
The laboratory should follow Good Laboratory Practice (GLP) and establish its own performance characteristics for QIAstat-Dx RP SARS-CoV-2 Control Panel in demonstrating adequate system performance.

2 Reviews
-
Excellent
The company is known for its excellent services and mannered communication with customers!
-
QIAstat-Dx RP SARS-CoV-2 Control Panel
QIAstat-Dx RP SARS-CoV-2 Control Panel is intended for use as an external positive and negative quality control to monitor the performance of in vitro laboratory nucleic acid testing procedures for the qualitative detection of bacterial and viral pathogens.