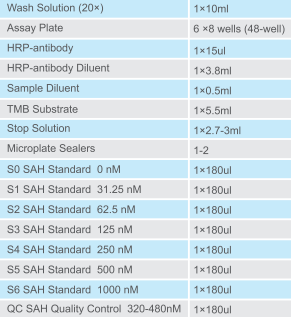
S-AdenosylHomocysteine (SAH) Elisa Kit, Large Range | IK00303
- SKU:
- ART-IK00303
- Availability:
- Usually Shipped in 5 Working Days
- Size:
- 48 Tests
Description
S-AdenosylHomocysteine (SAH) Elisa Kit, Large Range | IK00303 | Arthus Biosystems
Catalog No.
Ik00303
Packaging size
48 tests
Detection range
15.625 nM — 2000 nM
Target details
Methionine is an amino acid with sulfur methyl group, which is converted into S-adenosylmethionine (SAM) with ATP by methionine adenosyltransferase (EC 2.5.1.6). SAM is the sole methyl group donor for over 50 kinds of biologically active substances such as DNA, RNA, protein, phospholipids, hormones and neuro-transmitters, etc. After methyl group is transferred away from SAM under a mehtyltransferase, S- adenosylhomocysteine is formed, which is further metabolized into homocysteine (Hcy) after adenosine is removed. The homocysteine is metabolized to methionine through N5-methyl tetrahydrofolate methyltransferase (EC1.1.1.68) and coenzyme vitamin B12 catalytic pathway accepting methyl from N5-methyltetrahydrofolate. This is methionine cycle.
Methylation index is defined as the ratio of SAM and SAH. Methylation index is a better marker for methylation status and methylation capability.
SAH and Hcy connect the processes of providing methyl group to critical biological molecules and recycling methyl group from N-5- methyltetrahydrofolate. The levels of SAH will determine or reflect whether the methionine cycle is normal or not. Therefore, finding a way to better quantify them has valuable practical implications. Elevated SAH can cause damage to vascular endothelial cells, which is related to the levels of global genome methylation. SAH may be a potential biomarker for atherosclerosis. This immunoassay kit is used to measure the level of SAH in samples of soluble form.
Principle of the assay
This direct competitive ELISA (Enzyme Linked Immunosorbent assay) is designed to measure the level of S-adenosyl-L-methionine (SAH) in the sample. SAH conjugated with macromolecule is immobilized on the micro-titer plate. Standards and samples are pipetted into the wells, and then the HRP-conjugated antibody against SAH is added. The free SAH molecule in samples or standards competes with the immobilized SAH on the micro-titer plate surface for binding sites of the antibody. After discarding the mixed solution and washing each well, TMB substrate solution is added. The substrate solution turns blue under the effect of HRP (horseradish peroxidase), and changes into yellow once stop solution (acid) is added. The color develops in inverse proportion to the amount of SAH in the sample (or standards). The optical density of the remaining solution (0D450) is measured at 450nm using micro-plate spectrophotometer. The level of SAH in samples can be calculated through standard curve generated with standards.
Sample collection and storage
- Sample collection must be carried out at 4°C. Remove precipitation by centrifugation when necessary, and test sample as soon as possible or store at -20°C or below. Avoid repeated freeze-thaw cycles.
- Avoid NaN3 as it will inactivate HRP.
Procedure
- Rewarm all reagents by putting them to room temperature and mix them well. Take out appropriate number of wells and put the remaining wells back into the original sealing bag. Seal and store it at -20°C.
- Add 50u1 sample diluent (blank well), standards and sample into each well.
- Prepare HRP-antibody solution: Dilute HRP-antibody with HRP-antibody Diluent at 1:250, which should be used up within a week. Mix thoroughly and store in dark. Prepare the right amount that will be used in a week. Note: Always thoroughly centrifuge the HRP-antibody vial to recover all the 15-17u1 HRP-antibody. It is recommended to collect all samples and measure them together in 1-3 experiments in a week by using 3.8m1 of HRP-antibody Diluent to recover all HRP-antibody from the vial by washing it out several times.
- Add 50u1 HRP-conjugated antibody into each well except for the blank well.
- Put the plate onto oscillator and shake for a while to mix the reagents, and seal the plate with micro-plate sealer. Incubate the plate at 37°C for 1 hour.
- Peel the sealer carefully, and discard the remaining solution in the wells. Add at least 300u1 wash solution in each well and maintain this state for 30 seconds, then remove wash solution. Repeat these steps 3 times to finish washing process. Or use auto-washer instead.
- Add TMB Substrate and add 100u1 blending substrate to each well. Shake gently and seal the plate. Incubate the plate at 37°C for 15 minutes without light.
- Add 50u1 Stop Solution to end reaction.
- Measure absorbance of each well at 450 nm wavelength within 15 minutes. Set zero according to the blank well.
Data processing
Always clear the background by subtracting the absorbance at 450 nm (OD450) of blank well from that of the test well. The binging rate of each well (standard or sample) is equal to A/ASO, with A being the average absorbance of the standard wells or the sample wells, ASO being the average absorbance of the SO standard wells.
Creation of standard curve: Construct a standard curve by plotting the binding rate of each standard on the y-axis against the logarithm of its concentration on x-axis. Use quadratic polynomial curve to fit the data (r>0.99). The SAH level of the sample can be calculated by substituting its binding rate into the standard curve equation. Then multiply the extent of dilution if the sample has been diluted.
Note
- Using reagents and samples without warm-up or ambient temperature less than 20°C may lead to reduced OD450 reading.
- Extra drying of wells after washing may have negative effects on the results, such as poor standard curve and poor repeatability. To avoid it, perform the next step immediately after washing.
- Mix solution well and wash completely, as these procedures will have influence on the assay.
- Seal the plate with micro-plate sealer and avoid light when incubating the plate.
- Duplicate wells for standards and samples are recommended, and quadratic polynomial is suggested for standard curve fitting (R2>0.99). The detected concentration of quality control vial should be in the detection range.
- The concentrated wash solution may form crystals. Warm it up to allow salts to dissolve completely before diluting.
- The micro-plate sealers should be disposed after each use in order to avoid cross contaminations.
- The HRP substrate (TMB) should be kept in dark.
- The Stop Solution is diluted sulfuric acid. Avoid direct contact with skin and other things.
Storage and expiration
Storage condition: Except for the HRP substrate and stop solution that are stored at 2-8°C, all other ingredients and strips can be frozen stored.
Expiration: About 6 months from the date of shipment when storing it in refrigerator. If not planning to use it soon, users can put the assay plate, HRP-anti-SAM antibody, standards, HRP antibody and sample diluents in freezer for longer storage time or/and better results.
Components
Standard curve
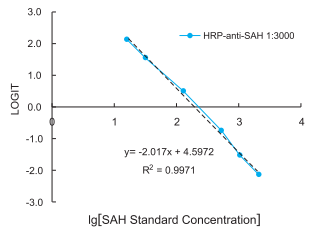
Figure 1: Standard curve HRP-anti-SAH antibody (clone 301-3) that was used at 1:3000 in a competitive ELISA. LOGIT = Ln(A/Aso /(1-A/Aso))






