AffiVET® Bird Flu H5 Antigen Rapid Test Card
- SKU:
- AFG-VGS-10
- Method:
- Rapid Test
- Size:
- 40 Tests/kit
- Species:
- Poultry
- Specimen:
- Secretions
Description
Avian influenza virus H5 (AIV-H5) antigen rapid test card
1. Introduction
Avian influenza hemagglutinin according to their outer membrane (H) and neuraminidase (N) protein antigen is divided into a plurality of different and different subtypes. H5 avian influenza virus antigen rapid test card is specifically designed to detect H5 subtypes of the virus detection reagent.
The AIV H5 Ag test strip is based on fast immuno-chromatography technique to detect AIV H5 Ag in birds trachea or cloaca (or feces). After adding sample, the sample move along with the colloidal gold labeling H5-AIV antibody, is there is H5-AIV antigen in the sample, it combines with antibody on T line and show wine-red color; if there is no H5-AIV antigen, then no color reaction.
2. Contents
| 1 | H5 Ag test card | 40 pieces |
| 2 | Swab | 40 pieces |
| 3 | Dropper | 40 pieces |
| 4 | Sample buffer | 40 bottles |
| 5 | Disposable gloves | 1 bag |
| 6 | Manual | 1 piece |
3. Test procedure
1) Use Swab to collect sample from cloaca (recommended) or trachea, the swab should be inserted into cloaca or trachea, wipe and rotate in inner well repeatedly. To young birds, it can use dropper to collect fresh feces directly.
2) Insert the swab into the tube with sample buffer immediately, rotate the swab strongly on tube well for at least 10 times and mix the solution evenly, making the sample been dissloved in sample buffer fully. Press the swab on tube well above the liquid level, making the liquid been pressed out fully, discard swab.
3) If the particles are big in feces, it may be static for 1-2 min, making bigger particles precipitate.
4) Take out test card from sealed bag. Put it on flat surface, use dropper to draw upper-layer bright liquid, drop 5 drops slowly, one by one, to the sample hole with “S” mark.
5) Put the test card at room temperature for 10-15min, read the result, the result is invalid after 15 minutes.
4.Results judgement
1) Negative: only control line is seen wine red;
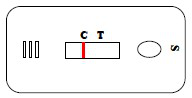 2) Positive: control line and test line are both seen wine red;
2) Positive: control line and test line are both seen wine red;
 3) Invalidation: control line isn’t seen wine red.
3) Invalidation: control line isn’t seen wine red.
 5. Note
5. Note
1) Use the sample buffer in the package. Do not mix use sample buffer from different supplier, different products. Never use Normal saline、PBS、Tap water、Pure water as sample buffer, because sample buffer from other sources will lead to false result(false positive or false negative result).
2) The sample buffer is not RNA enzyme clean level, so it may have false negative result if use the extracted sample for PCR test directly. If need to review by PCR, we recommend collect double sample at the same time, one for rapid test, one for PCR review test.
3) Do not use product with broken Aluminum foil bag package. Use test card in 1 hour after taking out.
4) The collected sample should be tested immediately, avoid long time storage.
5) The sample collection directly affect the accuracy of testing, so ensure collect sample correctly and fully.
6) Collect feces sample too much will affect the normal surge of liquid, and lead to wrong result (black area appear), and too little sample may lead to false negative result, so collect sample as following picture.
7) Avoid bubble when adding sample to test card.
8) The used test card should be handled as local pollution properly.
9) Do not use test card out of expiry date.
10) Deal with the used test card and sample as waste according to local regulation strictly.
6.Frequently Asked Questions
1) Limitation of the product
This kit is a qualitative screening reagent, can detect AIV H5 antigen accurately, but if sample is not enough or concentration of antigen is lower than sensitivity, it may appear negative result. A clear clinical diagnosis should be made after taking into account all clinical and laboratory phenomenon.
2) The sensitivity of the product.
The limit value to detect H5N1 Ag is 0.25HAU. But because different strains of avian influenza of the same subtype have antigenic drift, the minimum detectable amount of avian influenza strains tested for other H5 subtypes may be slightly less than 0.25 HAU. Meanwhile, the H5 avian influenza antigen RE-4 / RE-5 / RE-6 / RE-7 strain used for the hemagglutination inhibition test can not be used as a positive control. Because this series of antigens is produced by genetically engineered synthetic vaccine strains, their antigenicity is quite different from that of natural strains.
3) Thespecificity of the product.
The reaction with H1N1 / H2N2 / H3N2 / H7N3 / H9N2 strains have a negative representative. it has no cross-reaction with similar symptoms to those of fowl cholera, newcastle disease, infectious bronchitis
7. Storage and expiry date
Specifications: 40T/kit
Storage: Store at 2-30 ℃. Return to room temperature before open the Aluminum foil bags for cold storage.
Expiry date: 24 months; date of production is on box.
1 Review
-
faster and reliable results
This H5 avian influenza rapid test is a great addition to my clinic! It's quick, easy to use, and gives me reliable results on the spot. No more waiting for labs - perfect for catching outbreaks early.






