Description
The IMMUNO-TEK Bovine IgG ELISA Kit is a rapid, easy to use enzyme linked immunosorbent assay (ELISA) designed for the measurement of bovine IgG in bovine colostrum, milk, serum, plasma or other biological fluids. The assay contains mostly ready-to-use reagents and takes less than two hours to perform.
The IMMUNO-TEK Bovine IgG ELISA Kit is for Research Purposes Only.
Zeptometrix | 0801198 | Bovine IgG ELISA Datasheet
PRINCIPLE OF THE TEST
Microplate wells coated with polyclonal antibodies to bovine IgG form the capture phase of the assay. Captured bovine IgG then reacts with detector antibody, which is a polyclonal anti-bovine IgG, conjugated with horseradish peroxidase. Enzyme activity in the wells is then quantified using tetramethyl benzidine as a substrate.
REAGENTS
Materials Supplied:
• Microplate, (1x96 well): Strips coated with purified goat anti-bovine IgG
• Detector Antibody (12 mL): Contains conjugated goat anti-bovine IgG peroxidase
• Bovine IgG Standard (7 mL): Contains bovine IgG and Assay Diluent
• Assay Diluent (100 mL): Contains PBS, Triton X-100® and 2-chloroacetamide
• Plate Wash Buffer (125 mL): Contains PBS, Tween 20® and 2-chloroacetamide
• Substrate (12 mL): Contains Tetramethyl Benzidine (TMB)
• Stop Solution (12 mL): Proprietary formulation
• Microplate Sealers (1 pk): 10 sealers per pack
• Plastic Bag (1 bag): For storage of unused microplate strips
® Triton X-100 is a registered trademark of Union Carbide Chemicals and Plastics Co., Inc. Tween 20 is a registered trademark of Imperial Chemical Industries.
Materials required but not supplied:
• Disposable gloves
• Test tubes and racks for preparing specimen and IgG standard dilutions
• Validated adjustable micropipettes, single and multi-channel
• Distilled or deionized water
• Graduated cylinders and assorted beakers
• Validated microtiter plate reader
• Automatic microtiter plate washer or manual vacuum aspiration equipment
• Timer
STORAGE
Store all kit reagents at 2-8°C. Do not freeze. Unused microplate strips should be kept in a sealed bag with desiccant to minimize exposure to moisture. All reagents stored and handled properly are stable at least until the expiration date printed on the kit box label.
PRECAUTIONS
FOR RESEARCH USE ONLY. NOT FOR USE IN DIAGNOSTIC PROCEDURES.
• Prior to performing the assay, carefully read all instructions.
• Use universal precautions when handling kit components and test specimens.*
• To avoid cross-contamination, use separate pipette tips for each specimen.
• When testing potentially infectious specimens, adhere to all applicable local, state and federal regulations regarding the disposal of biohazardous materials.
• Stop Solution contains hydrochloric acid, which may cause severe burns. In case of contact with eyes or skin, rinse immediately with water and seek medical assistance. Wear protective clothing and eyewear.
*MMWR, June 24, 1988, Vol. 37, No. 24, pp. 377-382, 387-388
PREPARATION OF REAGENTS
Plate Wash Buffer
Dilute 10X Plate Wash Buffer 1:10 in distilled or deionized water prior to use. Mix thoroughly. Prepared 1X Plate Wash Buffer
can be stored at 2-8°C for up to one week. Additional 10X Plate Wash Buffer (ZMC Catalog #: 0801060) may be ordered if needed.
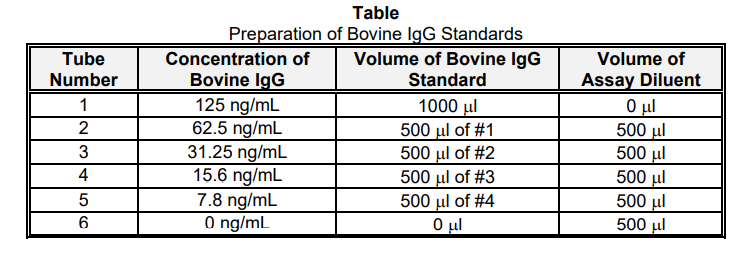
Bovine IgG Standard Curve
Label 6 test tubes as shown in Table. The Bovine IgG Standard is provided at 125 ng/mL. This should be diluted in Assay
Diluent as follows to prepare a standard curve.
SPECIMEN DILUTIONS
Bovine colostrum will normally contain 50-80 mg/mL of IgG antibody. A 1:1,000,000 dilution of bovine colostrum in Assay Diluent is recommended for initial testing.
Bovine milk will normally contain ~0.5 mg/mL of IgG antibody. A 1:10,000 dilution of bovine milk in Assay Diluent is recommended for initial testing.
Bovine serum will normally contain 25-30 mg/mL of IgG antibody. A 1:500,000 dilution of bovine serum in Assay Diluent is recommended for initial testing.
TEST PROCEDURE
Allow all reagents to reach room temperature before use. Label test tubes to be used for the preparation of standards and specimens. If the entire 96 well plate will not be used, remove surplus strips from the plate frame and place into the resealable Plastic Bag with desiccant. Seal bag and store at 2-8°C.
Step 1: Label each strip on its end tab to ensure identity should the strips become detached from the plate frame during the assay.
Step 2: Designate one well on the plate and leave empty. This well will serve as a substrate blank.
Step 3: Pipette 200 ul of standards #1-6 into duplicate wells.
Step 4: Pipette 200 ul of each specimen into duplicate wells.
Step 5: Cover the microplate with a plate sealer and incubate the plate for 30 minutes at room temperature.
Step 6: Aspirate the contents of each well and wash the wells 4 times with 1X Plate Wash Buffer. To wash, fill the wells with 300 ul of 1X plate wash buffer and aspirate. Perform 4 fill/aspirate cycles. After the final wash cycle, thoroughly blot the plate by carefully striking the plate on a pad of absorbent paper towels. Continue until no visible droplets of Plate Wash Buffer are observed.
Step 7: Pipette 100 ul of Detector Antibody into each standard and specimen well. Do not add Detector Antibody to the substrate blank well.
Step 8: Cover the plate with a plate sealer and incubate for 30 minutes at room temperature.
Step 9: Wash the plate 4 times with Plate Wash Buffer as described in Step 6.
Step 10: Pipette 100 ul of Substrate into each well including the substrate blank well.
Step 11: Incubate the plate for 30 minutes at room temperature. A blue color will develop in wells containing bovine IgG.
Step 12: Pipette 100 ul of Stop Solution into each well. A color change from blue to yellow will occur.
Step 13: Within 15 minutes, read the optical density of each well at 450 nm using a microtiter plate reader.
CALCULATION AND INTERPRETATION OF RESULTS
1. Calculate the mean optical density (OD) values for each set of standards and diluted specimen samples.
2. Using linear graph paper or graphing software, plot the mean OD values of each standard on the Y-axis versus the corresponding concentration of standards on the X-axis.
3. Draw the best fit curve through these points to construct a standard curve. In most cases a linear regression plot will be sufficient but for the most accurate results use a point-to-point or 4-parameter logistic regression.
4. Determine the IgG concentration of each diluted specimen sample by interpolation from the standard curve.
5. Multiply by the dilution factor used to dilute each specimen sample to determine the IgG concentration of the original specimen sample.
Test Validity:
• For the test to be valid, the mean optical density of the 0 ng/mL standard and the substrate blank must be below 0.200.
• The mean optical density of the 125 ng/ml must be above 1.000.
• It is recommended that specimen samples be re-assayed with a greater or lesser dilution when the mean optical density value does not fall within the standard curve.
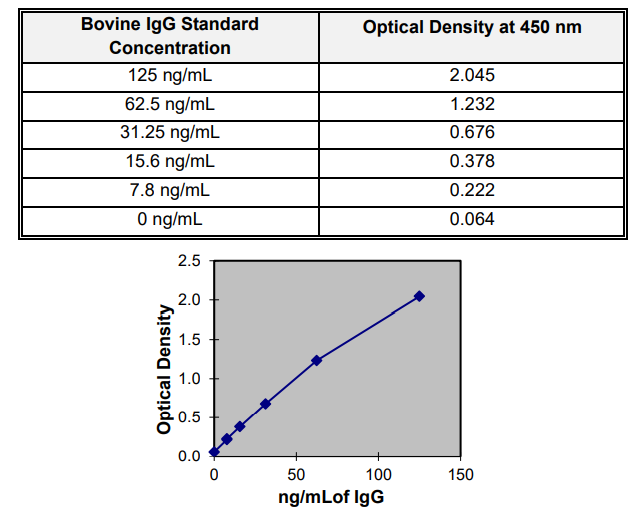
EXPECTED RESULTS
Below is an example of a standard curve and should not be used to calculate actual samples. Variations may be observed
from laboratory to laboratory due to pipetting, incubator temperatures, plate readers, etc.
PROCEDURAL FLOW CHART
PREPARE REAGENT DILUTIONS
=>
PIPETTE SPECIMENS AND STANDARDS
=>
INCUBATE 30 MINUTES AT ROOM TEMPERATURE
=>
WASH PLATE
=>
PIPETTE DETECTOR ANTIBODY
=>
INCUBATE 30 MINUTES AT ROOM TEMPERATURE
=>
WASH PLATE
=>
PIPETTE SUBSTRATE SOLUTION
=>
INCUBATE 30 MINUTES AT ROOM TEMPERATURE
=>
ADD STOP SOLUTION AND READ AT 450 NM
FOR RESEARCH USE ONLY. NOT FOR USE IN DIAGNOSTIC PROCEDURES
3 Reviews
-
Fast delivery
the services they provided and their dealing with their customers is totally perfect.
-
Originale
come il loro nome Genatur i loro prodotti sono totalmente biologici. Nessun uso di sostanze chimiche nocive davvero soddisfatto dei prodotti.
-
es bueno
mi experiencia con Genatur es muy buena. Estoy enamorada de sus productos y servicios.







![HEV IgG ELISA (ELISA Kit) [CE] HEV IgG ELISA (ELISA Kit) [CE]](https://cdn11.bigcommerce.com/s-ryt90hjx0j/images/stencil/590x590/products/38642/55299/logo-diasource__50886.1773239006.png?c=1)