NATtrol Norovirus GI/GII Positive Control
- SKU:
- MDZ049 --- NATNOV-6MC-IVD
- Performance:
- Cepheid® GeneXpert® Platform
- OLD CAT#:
- MDZ049 [Will be available until existing lots are consumed]
- NEW CAT#:
- NATNOV-6MC-IVD [New reference for identical product]
Description
NATtrol™ Norovirus GI/GII Positive Control (MDZ049) is an in vitro diagnostic external run control intended for use in evaluating and monitoring of qualitative molecular diagnostic assays for the detection of Norovirus GI/GII nucleic acid. The routine and repetitive use of external run controls enables laboratories to monitor daily test variation, lot-to-lot test kit performance, individual operator variation, and can provide assistance in identifying increases in random or systemic error. The NATtrol™ Norovirus GI/GII Positive Control contains intact organisms and should be run in a manner identical to that used for clinical specimens.
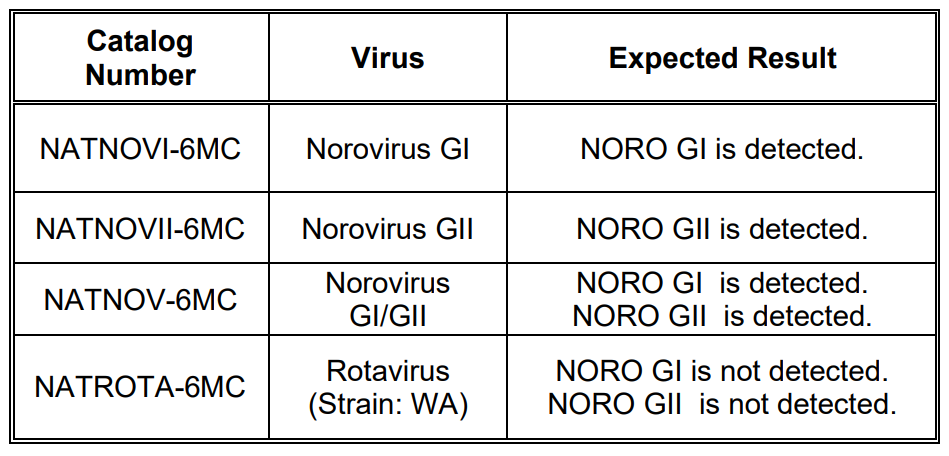
NATtrol™ Norovirus External Run Controls (NATNOVI-6MC, NATNOVII-6MC, NATNOV-6MC and NATROTA-6MC)* are formulated with purified, intact virus particles that have been chemically modified to render them non-infectious and refrigerator stable. Each control contains 6 x 0.125 mL vials of NATtrol™ Norovirus GI, NATtrol™ Norovirus GII, a blend of NATtrol Norovirus GI/GII or NATtrolTM Rotavirus. These controls aresupplied in a purified protein matrix that mimics the composition ofa true clinical specimen.
INTENDED USE:
NATtrol™ Norovirus GI/GII Positive Control (MDZ049) is an in vitro diagnostic external run control intended for use in evaluating and monitoring of qualitative molecular diagnostic assays for the detection of Norovirus GI/GII nucleic acid. The routine and repetitive use of external run controls enables laboratories to monitor daily test variation, lot-to-lot test kit performance, individual operator variation, and can provide assistance in identifying increases in random or systemic error.
The NATtrol™ Norovirus GI/GII Positive Control contains intact organisms and should be run in a manner identical to that used for clinical specimens.
NATtrol™ Norovirus External Run Controls are designed to evaluate the performance of nucleic acid tests for determination of the presence of Norovirus nucleic acids. NATNOVI-6MC, NATNOVII-6MC, NATNOV-6MC and NATROTA-6MC enables laboratories to monitor test variation, lot-to-lot test kit performance, operator variation and can provide assistance in identifying random or systemic error.
PRODUCT SUMMARY AND EXPLANATION:
NATtrol™ Norovirus GI/GII Positive Control is formulated with purified, intact organisms that have been chemically modified to render them non-infectious and refrigerator stable*.
Each NATtrol™ Norovirus GI/GII Positive Control contains 6 x 0.5 mL vials of NATtrolTM Norovirus GI/GII formulated in a Purified Protein Matrix that is fully commutable with true clinical specimens.
PRINCIPLE:
NATtrol™ Norovirus GI/GII Positive Control contains Norovirus GI and Norovirus GII inactivated by ZeptoMetrix’s patented NATtrol™ process formulated in a proprietary Purified Protein Matrix that mimics the composition of a true clinical specimen. These are full process controls designed to monitor the effectiveness of extraction, amplification, and detection in nucleic acid testing procedures. These controls are suitable for use in in-house molecular assays and commercially available platforms.
PRECAUTIONS:
NOT FOR USE IN HUMANS:
These products are NOT intended for use in the manufacture or processing of injectable products subject to licensure under the USA Food and Drug Administration Section 351 of the Public Health Service Act or for any other product intended for administration to humans.
RECOMMENDED STORAGE:
NATtrol™ Norovirus GI/GII Positive Control should be stored at 2-8°C. When stored as directed, controls are suitable for use for up to 56 days (8 weeks) once opened.
INSTRUCTIONS FOR USE:
Vortex NATtrol™ Norovirus GI/GII Positive Control vials for 10 seconds to mix.
Follow the manufacturer instructions for use as a clinical sample.
LIMITATIONS:
NATtrol™ Norovirus GI/GII Positive Control is a USA FDA Class 1 exempt, unassayed, in vitro diagnostic external run control and is for professional use only. NATtrol™ Norovirus GI/GII Positive Control is not intended for use as a substitute for the internal controls provided by in vitro diagnostic kit manufacturers. Quality control materials should be used in accordance with local, state, federal and accreditation requirements.
EXPECTED RESULTS:
NATtrol™ Norovirus GI/GII Positive Control tested positive for
Norovirus GI and Norovirus GII in the Xpert® Norovirus Assay.
Each laboratory must evaluate the controls and establish their own acceptance criteria.
Although the NATtrol™ Norovirus GI/GII Positive Control contains inactivated microorganisms, handling and disposal should be conducted as if potentially infectious.
This control contains material of human and animal origin and the user should observe Universal Precautions when handling and disposing of this product. Disposal must follow local regulations if more stringent then regulations enforced by the CDC or the FDA.
Do not pipette by mouth. To avoid cross-contamination, use separate transfer pipettes or tips for all materials.
Do not use beyond the expiration date shown on the label. If product is received damaged or leaking, contact ZeptoMetrix LLC for instructions.
ETIOLOGIC STATUS/BIOHAZARD TESTING:
NATtrol™ inactivation was completed on the stocks used to formulate each control and further verified by the absence of viral growth in a validated tissue culture-based infectivity assay.
The Purified Protein Matrix used in the manufacture of this product is treated with 0.09% sodium azide. It was manufactured from materials that have been tested and found non-reactive at the donor level for HIV-1/HIV-2 Antibody, HBsAg and HCV Antibody by FDA licensed donor screening test methods. All materials are also tested for HIV-1 and HCV by FDA approved Nucleic Acid Test (NAT) methods. Heat inactivated bovine based source materials used in the manufacture of this product meet applicable USDA requirements for abattoir sourced animals, traceability and country of origin. The materials were collected at USDA licensed establishments or legally imported from countries recognized by the USDA as negligible or controlled for risk for Bovine Spongiform Encephalopathy (BSE) and other exotic disease agents. Donor animals were inspected ante and post mortem at the abattoir as required by the USDA.
PRODUCT WARRANTY:
ZeptoMetrix LLC’s limited product warranty and other terms and conditions related to the purchase and use of ZeptoMetrix products are set fo rth in ZeptoMetrix’s Terms and Conditions of sale found on ZeptoMetrix’s website at Sales Terms and Conditions.
DISCLAIMER AND LIMITATION OF LIABILITY:
ZeptoMetrix LLC disclaims all warranties with respect to this document and the information contained herein, expressed or implied, including but not limited to those of merchantability, fitness for a particular purpose, or non-infringement. To the extent allowed by law, in no event shall ZeptoMetrix LLC be liable, whether in contract, tort, warranty, or consequential damages or lost profits in connection with or arising from this document and the information contained herein, including but not limited to the use thereof even if ZeptoMetrix is advised of the possibility of such damages.
3 Reviews
-
amazing product
It is an amazing product, I have used it and the results I have seen are quite good.
-
producto increíble
Este producto es increíble, lo he usado y este producto tiene resultados muy efectivos.
-
perfect
It was very fast to answer my question about my order, which surprised me for such a fast delivery. Thank you, very much.






