Recombinant Core Streptavidin 2 (r-cSA2)
- SKU:
- GEN-NRPA18S
- Formulation:
- Lyophilized from 5mM PB (4mM Na2HPO4, 1mM NaH2PO4, pH7.4)
- Mol. Wt .:
- 13.3 kDa subunit; Tetramer 53.2 kDa
- Theory pI:
- 6.09
- Theoretical activity:
- 18.3 protein u / mg (r-cSA2: Biotin = 1: 4 (mol: mol))
- Resources:
- Escherichia coli (E. coli)
- Purity:
- ≥95% by SDS-PAGE analysis
- Test activity:
- ≥16 protein u / mg (determined by NUPTEC according to the modified green method)
- Storage state:
- -20 °C
- Storage period:
- 3 years
Description
Streptavidin is a homotetrameric protein found in the culture broth of Streptomyces avidini. Similar to avidin, a streptavidin mole can bind 4 biotin moles with high affinity virtually unmatched in nature. Streptavidin lacks the carbohydrate side chains found on avidin and has an isoelectric point near neutrality. Therefore, it has a reduced non-specific binding level compared to avidin. Streptavidin has been widely applied in various biological fields, such as ELISA, IHC, TRFIA, PCR quantification, single-stranded nucleotide isolation, biomolecule purification, and monoclonal antibody production.
Compared to native streptavidin, r-cSA2 is improved in stability and solubility by removal of the activity-unrelated sequence. Compared to r-csa (nrpa09), the amino acid sequence of this product does not contain cysteine, so it does not need reduction therapy.
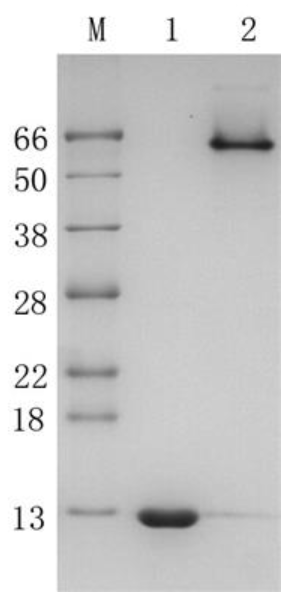
M: Protein molecular weight marker
Lane 1: r-cSA2 subunit
Channel 2: r-cSA2 homotetramer
For research use only!