Description
OptiPrep™ is a sterile, endotoxin-tested, multi-purpose separation medium of 60% iodixanol in water with a density of 1.32 g/ml, ideal for isolating viruses, subcellular organelles, membranes, macromolecules and cells, especially when controlling osmolality is a priority. Iodixanol has undergone rigorous clinical testing and is manufactured under strict EU cGMP compliance.
OptiPrep™ The optimum density gradient medium
OptiPrep™ The ideal density gradient medium for purification and analysis of viruses
Specifications
- Iodixanol 60% (w/v)
- Density 1.320 ± 0.001 g/ml
- Osmolality ~170 mOsm
- Endotoxin < 1.0 EU/ml
Applications
- Membrane trafficking and cell signaling analysis
- Endocytosis and exocytosis analysis
- Mammalian and non-mammalian cell purification; routinely achieve maximum yield of non-human lymphocytes with no neutrophil contamination
- Mammalian and nonmammalian organelle purification
- Plasma membrane and domain purification
- Membrane vesicle and cytosol purification
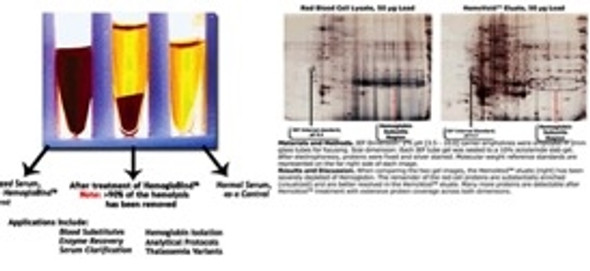
- Virus purification
- Plasma lipoprotein purification
- Protein and protein complex purification
- Plasmid DNA purification
- Ribonucleoprotein purification
Features and properties
- Developed as an X-ray contrast medium and has therefore been subjected to rigorous clinical testing.
- Non-ionic
- Non-toxic to cells
- Metabolically inert.
- Biologically and physiochemically very similar to Nycodenz® with some key differences:
- Structurally, an approximate dimer of Nycodenz®
- Osmolality approximately half that of a Nycodenz® solution of the same density
- Can be made iso-osmotic at all densities
- Can be added directly to a cell or organelle suspension without changing its osmolality, facilitating purification in flotation gradients
- Forms self-generated gradients more quickly than Nycodenz® due to higher molecular mass
- High viability of gradient-purified mammalian and non-mammalian cells
- Effective organelle and membrane vesicle fractionation using either pre-formed or self-generated gradients
- Virus infectivity at least 100x higher than from CsCl gradients
- Better preservation of protein-protein and protein-nucleic acid complexes than with sucrose or glycerol gradients
- Plasma lipoproteins analyzed in self-generated gradients within 4h
- Endotoxin level less guaranteed to be less than 1.0 EU/ml (as determined by limulus amebocyte lysate (LAL) test); however, our goal is to produce batches with endotoxin levels lower or equal to 0.13 EU/ml. Actual density and endotoxin level values for each batch are specified in Certificates of Analysis.
- Sterility is claimed according to Ph. Eur.
- Manufactured, packaged and released in compliance with:
- Current EU Guide to Good Manufacturing Practice
- Fresenius Kabi AS Quality System
- Fresenius Kabi AS Manufacturing License
4 Reviews
-
Efficiency
I recently used OptiPrep for density gradient centrifugation in my cell isolation experiments, and I’m really impressed! The product delivered excellent separation of cell populations, allowing me to obtain high-purity fractions with minimal contamination. The protocol was easy to follow, and the results were consistent across multiple runs. OptiPrep is definitely a reliable choice for anyone working in cell biology or biochemistry. Highly recommended for efficient and effective cell separation!
-
Highly recommend OptiPrep
This sterile, endotoxin-tested medium is fantastic for isolating viruses, organelles, membranes, and more. Plus, it allows for precise control of osmolality, making it ideal for sensitive experiments.
-
higher quality
I ordered the product. Its results are beyond my expectations. Feeling very happy after using this product
-
Eccellente
per mantenere nel mercato la cosa più importante è mantenere la qualità. Devo dire che Gentaur è il migliore nel mantenere una qualità superiore rispetto ad altre aziende produttrici di antibiotici