PCR
PCR Animation
If the nucleotide sequences at the ends of a particular DNA region are known, the intervening fragment can be amplified directly by the polymerase chain reaction (PCR). Here we describe the basic PCR technique and three situations in which it is used.
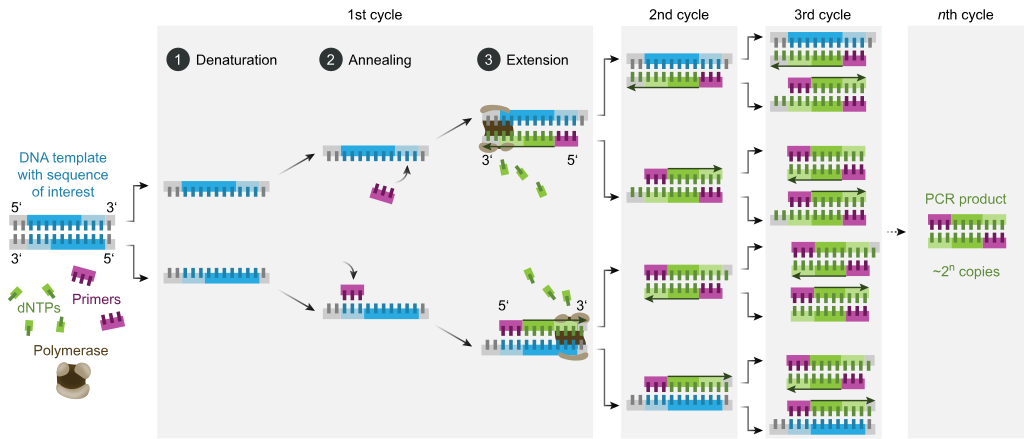
The PCR depends on the ability to alternately denature (melt) double-stranded DNA molecules and renature (anneal) complementary single strands in a controlled fashion. As in the membrane-hybridization assay described earlier, the presence of noncomplementary strands in a mixture has little effect on the base pairing of complementary single DNA strands or complementary regions of strands. The second requirement for PCR is the ability to synthesize oligonucleotides at least 18–20 nucleotides long with a defined sequence. Such synthetic nucleotides can be readily produced with automated instruments based on the standard reaction scheme.
A typical PCR procedure begins by heat-denaturation of a DNA sample into single strands. Next, two synthetic oligonucleotides complementary to the 3' ends of the target DNA segment of interest are added in great excess to the denatured DNA, and the temperature is lowered to 50–60 C. These specific oligonucleotides, which are at a very high concentration, will hybridize with their complementary sequences in the DNA sample, whereas the long strands of the sample DNA remain apart because of their low concentration. The hybridized oligonucleotides then serve as primers for DNA chain synthesis in the presence of deoxynucleotides (dNTPs) and a temperature-resistant DNA polymerase such as that from Thermus aquaticus (a bacterium that lives in hot springs).
This enzyme, called Taq polymerase, can remain active even after being heated to 95 C and can extend the primers at temperatures up to 72 C. When synthesis is complete, the whole mixture is then heated to 95 C to melt the newly formed DNA duplexes. After the temperature is lowered again, another cycle of synthesis takes place because excess primer is still present. Repeated cycles of melting (heating) and synthesis (cooling) quickly amplify the sequence of interest. At each cycle, the number of copies of the sequence between the primer sites is doubled; therefore, the desired sequence increases exponentially—about a million-fold after 20 cycles—whereas all other sequences in the original DNA sample remain unamplified.
- Product
- Qty in Cart
- Quantity
- Price
- Subtotal
-

Hemochromatosis C282Y Kit | S01193-50-T
£848.85Hemochromatosis C282Y Kit | S01193-50-Tfrom Sacace Biotechnologies is available for delivery. RT PCR test for detection of Cys282TyrStorage & Shipping :ON REQUEST441-S01193-50-T£848.85 -

Hemochromatosis S65C Kit | S01192-50-T
£848.85Hemochromatosis S65C Kit | S01192-50-Tfrom Sacace Biotechnologies is available for delivery. RT PCR test for detection of Ser65CysStorage & Shipping :ON REQUEST441-S01192-50-T£848.85 -

Hemochromatosis H63D Kit | S01191-50-T
£848.85Hemochromatosis H63D Kit | S01191-50-Tfrom Sacace Biotechnologies is available for delivery. RT PCR test for detection of His63AspStorage & Shipping :ON REQUEST441-S01191-50-T£848.85 -

Hemochromatosis Genotyping Kit NEW | HM-1-50-T
£1,178.32Hemochromatosis Genotyping Kit NEW | HM-1-50-Tfrom Sacace Biotechnologies is available for delivery. RT PCR test for detection of His63Asp, Ser65Cys, Cys282TStorage & Shipping :ON REQUEST441-HM-1-50-T£1,178.32 -

CFTR Genetics RT-PCR NEW | T01005-96
£848.85CFTR Genetics RT-PCR NEW | T01005-96from Sacace Biotechnologies is available for delivery. F508del, G542X, W1282X, N1303K, 2143delT, 2184insA, 3849+10kb C>T, dele2,3 (21kb), 2184insA, 3821delT, R334W, L138ins, 394delTT, S1196X, 604insA, 3944delTG,...441-T01005-96£848.85 -

Chicken & Turkey RT-PCR NEW | HM-1-50-T-1
£922.06Chicken & Turkey RT-PCR NEW | HM-1-50-T-1from Sacace Biotechnologies is available for delivery. Real Time PCR kit for qualitative detection of the Chicken & Turkey Storage & Shipping : ON REQUEST441-HM-1-50-T-1£922.06 -


Halal Food RT-PCR | HM-1-50-T-2
£793.94Halal Food RT-PCR | HM-1-50-T-2 from Sacace Biotechnologies is available for delivery. Real Time PCR kit for qualitative detection of porcine genomic DNA. Storage & Shipping : ON REQUEST441-HM-1-50-T-2£793.94 -

CYP3A5*3 (G6986A) SNP-Screen | T01331
£848.85 - £1,146.29CYP3A5*3 (G6986A) SNP-Screen | T01331from Sacace Biotechnologies is available for delivery. RT-PCR test for detection of CYP3A5 gene mutation (rs776746) Available Formats: - Ready to Use 0,2 mL Tube - Ready to Use 12 x 8 Strip Storage & Shipping...441-T01331£848.85 - £1,146.29 -

CYP2C9*2 (C430T) SNP-Screen | T01104
£848.85 - £1,146.29CYP2C9*2 (C430T) SNP-Screen | T01104from Sacace Biotechnologies is available for delivery. RT-PCR test for detection of CYP2C9 gene mutation (Ile359Leu; rs1057910) Available Formats: - Ready to Use 0,2 mL Tube- Ready to Use 12 x 8 StripStorage &...441-T01104£848.85 - £1,146.29 -

CYP2C9*3 (A1075C) SNP-Screen | T01111
£848.85 - £1,146.29CYP2C9*3 (A1075C) SNP-Screen | T01111from Sacace Biotechnologies is available for delivery. RT-PCR test for detection of CYP2C9 gene mutation (Ile359Leu; rs1057910) Available Formats: - Ready to Use 0,2 mL Tube - Ready to Use 12 x 8 Strip Storage &...441-T01111£848.85 - £1,146.29 -

VKORC1 (?1173T) SNP-Screen | T01144
£848.85 - £1,146.29VKORC1 (?1173T) SNP-Screen | T01144from Sacace Biotechnologies is available for delivery. RT-PCR test for detection Vitamin K epoxide reductase complex subunit 1 gene mutation, Warfarin sensitivity (rs9934438) Available Formats: - Ready to Use 0,2 mL...441-T01144£848.85 - £1,146.29 -

VKORC1 (G3730A) SNP-Screen | T01145
£848.85 - £1,146.29VKORC1 (G3730A) SNP-Screen | T01145from Sacace Biotechnologies is available for delivery. RT-PCR test for detection Vitamin K epoxide reductase complex subunit 1 gene mutation, Warfarin resistance (rs7294) Available Formats: - Ready to Use 0,2 mL...441-T01145£848.85 - £1,146.29 -

PPARG2 (C34G) SNP-Screen | T01359
£848.85 - £1,146.29PPARG2 (C34G) SNP-Screen | T01359from Sacace Biotechnologies is available for delivery. Real Time PCR test for detection of Peroxisome Proliferator-Activated Receptor-Gamma-2 gene mutation (Pro12Ala, rs1801282) Available Formats: - Ready to Use 0,2 mL...441-T01359£848.85 - £1,146.29 -

ADRB2 (C5318G) SNP-Screen | T01358
£848.85 - £1,146.29ADRB2 (C5318G) SNP-Screen | T01358from Sacace Biotechnologies is available for delivery. RT-PCR test for detection adrenoceptor beta 2, surface gene (Gln27Glu, rs1042714) Available Formats: - Ready to Use 0,2 mL Tube - Ready to Use 12 x 8 Strip Storage...441-T01358£848.85 - £1,146.29 -

ADRB3 (T190C) SNP-Screen | T01360
£848.85 - £1,146.29ADRB3 (T190C) SNP-Screen | T01360from Sacace Biotechnologies is available for delivery. RT-PCR test for detection of SLCO1B1 gene mutation (Val174Ala; rs4149056) Available Formats: - Ready to Use 0,2 mL Tube - Ready to Use 12 x 8 Strip Storage &...441-T01360£848.85 - £1,146.29 -

FABP2 (A163G) SNP-Screen | T01361
£848.85 - £1,146.29FABP2 (A163G) SNP-Screen | T01361from Sacace Biotechnologies is available for delivery. RT-PCR test for detection of Fatty Acid Binding Protein 2 gene (Ala54Thr, rs1799883) Available Formats: - Ready to Use 0,2 mL Tube - Ready to Use 12 x 8...441-T01361£848.85 - £1,146.29 -

SLCO1B1 (T37041C) SNP-Screen | T01303
£848.85 - £1,146.29SLCO1B1 (T37041C) SNP-Screen | T01303from Sacace Biotechnologies is available for delivery. RT-PCR test for detection of SLCO1B1 gene mutation (Val174Ala; rs4149056) Available Formats: - Ready to Use 0,2 mL Tube - Ready to Use 12 x 8 Strip Storage...441-T01303£848.85 - £1,146.29 -

CYP2C19*2 (G681A) SNP-Screen | T01323
£848.85 - £1,146.29CYP2C19*2 (G681A) SNP-Screen | T01323from Sacace Biotechnologies is available for delivery. Available Formats: - Ready to Use 0,2 mL Tube - Ready to Use 12 x 8 Strip Storage & Shipping : ON REQUEST CYP2C19*2 (G681A) SNP-Screen T01323...441-T01323£848.85 - £1,146.29 -

COMT Val158Met SNP-Screen RT-PCR New | T01352-50-T
£848.85COMT Val158Met SNP-Screen RT-PCR New | T01352-50-Tfrom Sacace Biotechnologies is available for delivery. Real Time PCR kitStorage & Shipping :ON REQUEST441-T01352-50-T£848.85 -

CYP2C19*3 (G636A) SNP-Screen | T01324
£848.85 - £1,146.29CYP2C193 (G636A) SNP-Screen | T01324 *from Sacace Biotechnologies is available for delivery Description: RT-PCR test for detection of CYP2C193) Available Formats: - Ready to Use 0,2 mL Tube - Ready to Use 12 x 8 Strip Storage & Shipping : ON...441-T01324£848.85 - £1,146.29 -

PeriodontScreen RT-PCR (CE) | T01707-96-S
£848.85PeriodontScreen RT-PCR | T01707-96-S from Sacace Biotechnologies is available for delivery Real Time PCR test for detection and quantification of Porphyromonas endodontalis, Porphyromonas gingivalis, Aggregatibacter actinomycetemcomitans, Treponema...441-T01707-96-S£848.85 -

HSV I/II Typing RT-PCR (CE) | V38-FRT
£846.56 - £1,086.80HSV I/II Typing RT-PCR | V38-FRT from Sacace Biotechnologies is available for delivery Real Time PCR kit for qualitative detection and differentiation of Herpes Viruses I and II Storage & Shipping : The kit can be shipped at 2-8°C for 3-4 days...441-V38-FRT£846.56 - £1,086.80 -

Influenza A B RT-PCR (CE) | V36-FRT
£846.56 - £1,086.80Influenza A B RT-PCR (CE) | V36-FRT from Sacace Biotechnologies is available for delivery Real Time Amplification test for the detection of Influenza A and B Viruses Storage & Shipping : The kit can be shipped at 2-8°C for 3-4 days but should be...441-V36-FRT£846.56 - £1,086.80 -

HPV High Risk Screen RT-PCR Quant | V31-2FRT
£846.56 - £1,086.80HPV High Risk Screen RT-PCR Quant (CE) | V31-2FRT from Sacace Biotechnologies is available for delivery Real Time PCR kit for quantitative detection of Human Papillomavirus (16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59) Storage & Shipping : The kit...441-V31-2FRT£846.56 - £1,086.80 -

HHV7 RT-PCR Quant | V17-FRT
£846.56 - £1,086.80HHV7 RT-PCR Quant | V17-FRT from Sacace Biotechnologies is available for delivery Real Time PCR kit for quantitative detection of Human Herpes Virus 7 Storage & Shipping : The kit can be shipped at 2-8°C for 3-4 days but should be stored at...441-V17-FRT£846.56 - £1,086.80 -

HHV6 RT-PCR Quant (CE) | V10-FRT
£846.56 - £1,086.80HHV6 RT-PCR Quant | V10-FRT from Sacace Biotechnologies is available for delivery Real Time PCR kit for quantitative detection of Human Herpes Virus 6 Storage & Shipping : The kit can be shipped at 2-8°C for 3-4 days but should be stored at...441-V10-FRT£846.56 - £1,086.80 -

HSV I/II RT-PCR (CE) | V8-100FRT
£846.56 - £1,086.80HSV I/II RT-PCR (CE) | V8-100FRT from Sacace Biotechnologies is available for delivery Real Time PCR kit for qualitative detection of Herpes Viruses I and II Storage & Shipping : The kit can be shipped at 2-8°C for 3-4 days but should be stored...441-V8-FRT£846.56 - £1,086.80 -

HIV RT-PCR Qual | R-V0-FRT
£846.56 - £1,086.80HIV RT-PCR Qual (CE) | R-V0-FRT from Sacace Biotechnologies is available for delivery Real Time PCR kit for qualitative detection of Human Immunodeficiency Virus (HIV) Storage & Shipping : The kit can be shipped at 2-8°C for 3-4 days but should...441-R-V0-FRT£846.56 - £1,086.80 -

Gardnerella vaginalis/Lactobacillus species RT-PCR Quant (CE) | R-B7-FRT
£846.56 - £1,086.80Gardnerella vaginalis/Lactobacillus species RT-PCR Quant (CE) | R-B7-FRT from Sacace Biotechnologies is available for delivery Real Time Kit PCR kit for quantitative detection of Gardnerella vaginalis and Lactobacillus spp. Storage & Shipping : The...441-R-B7-FRT£846.56 - £1,086.80 -

Leishmania spp. RT-PCR | N3-FRT
£846.56 - £1,086.80Leishmania spp. RT-PCR (CE) | N3-FRT from Sacace Biotechnologies is available for delivery Real Time PCR kit for qualitative detection of Leishmania spp. Storage & Shipping : The kit can be shipped at 2-8°C for 3-4 days but should be stored at...441-N3-FRT£846.56 - £1,086.80 -

Candidosis RT-PCR Quant (CE) | F5-FRT
£846.56 - £1,086.80Candidosis RT-PCR Quant (CE) | F5-FRT from Sacace Biotechnologies is available for delivery Multiplex Real Time PCR test for the quantitative detection of Candida albicans, Candida glabrata, Candida krusei, Candida parapsilosis and Candida tropicalis...441-F5-FRT£846.56 - £1,086.80 -

Candida albicans/Candida glabrata /Candida krusei RT-PCR (CE) | F3-FRT
£846.56 - £1,086.80Candida albicans/Candida glabrata /Candida krusei RT-PCR (CE) | F3-FRT from Sacace Biotechnologies is available for delivery Multiplex Real Time PCR test for the qualitative detection of Candida albicans, Candida glabrata, and Candida krusei Storage...441-F3-FRT£846.56 - £1,086.80 -

HSV I /HSV II / T. pallidum RT-PCR | B87-FRT
£846.56 - £1,086.80HSV I /HSV II / T. pallidum RT-PCR (CE) | B87-FRT from Sacace Biotechnologies is available for delivery Multiplex Real Time PCR kit for qualitative detection of HSV I, HSV II and Treponema pallidum Storage & Shipping : The kit can be shipped at...441-B87-FRT£846.56 - £1,086.80 -

Ureaplasma parvum/Ur.urealyticum/ M.hominis RT-PCR Quant (CE) | B75-FRT Q
£846.56 - £1,086.80Ureaplasma parvum/Ur.urealyticum/ M.hominis RT-PCR Quant (CE) | B75-FRT Q from Sacace Biotechnologies is available for delivery Real Time PCR Kit for quantitative detection of Ureaplasma parvum, Ureaplasma urealyticum, Mycoplasma hominis Storage &...441-B75-FRT-Q£846.56 - £1,086.80 -

Bacterial Vaginosis RT-PCR Quant (CE) | B74-FRT
£846.56 - £1,086.80Bacterial Vaginosis RT-PCR Quant (CE) | B74-FRT from Sacace Biotechnologies is available for delivery Multiplex Real Time PCR kit for quantitative detection of Gardnerella vaginalis, Atopobium vaginae, Lactobacillus spp. and total bacteria...441-B74-FRT£846.56 - £1,086.80 -

Cronobacter sakazakii RT-PCR (CE) | B58-FRT
£846.56 - £1,086.80Cronobacter sakazakii RT-PCR (CE) | B58-FRT from Sacace Biotechnologies is available for delivery Real Time PCR Kit for detection of Cronobacter sakazakii Storage & Shipping : The kit can be shipped at 2-8°C for 3-4 days but should be stored at...441-B58-FRT£846.56 - £1,086.80 -

Leptospira 16s RNA RT-PCR (CE) | B49-FRT
£846.56 - £1,086.80Leptospira 16s RNA RT-PCR (CE) | B49-FRT from Sacace Biotechnologies is available for delivery Real Time Kit for the qualitative detection of Leptospira 16s RNA Storage & Shipping : The kit can be shipped at 2-8°C for 3-4 days but should be...441-B49-FRT£846.56 - £1,086.80 -

A.I.I. Screen RT-PCR (CE) | B45-FRT
£846.56 - £1,086.80A.I.I. Screen RT-PCR (CE) | B45-FRT from Sacace Biotechnologies is available for delivery Real Time PCR test for the qualitative detection and differentiation of Shigella Spp. E.coli, Salmonella spp., Campylobacter spp., Adenovirus F, Rotavirus A,...441-B45-FRT£846.56 - £1,086.80 -

Shigella/Salmonella/Campylobacter RT-PCR (CE) | B44-FRT
£846.56 - £1,086.80Shigella/Salmonella/Campylobacter RT-PCR | B44-FRT from Sacace Biotechnologies is available for delivery Real Time PCR Kit for detection and differentiation of Shigella spp and Enteroinvasive E.Coli (EIEC)/ Salmonella spp/Campylobacter spp Storage &...441-B44-FRT£846.56 - £1,086.80 -

Borrelia burgdorferi RT-PCR (CE) | B37-FRT
£846.56 - £1,086.80Borrelia burgdorferi RT-PCR | B37-FRT from Sacace Biotechnologies is available for delivery Real Time PCR Kit for qualitative detection of Borrelia burgdorferi in biological materials Storage & Shipping : The kit can be shipped at 2-8°C for 3-4...441-B37-FRT£846.56 - £1,086.80 -

Listeria monocytogenes RT-PCR Quant (CE) | B14-FRT
£846.56 - £1,086.80Listeria monocytogenes RT-PCR Quant | B14-FRT from Sacace Biotechnologies is available for delivery Real Time PCR Kit for quantitative detection of Listeria monocytogenes Storage & Shipping : The kit can be shipped at 2-8°C for 3-4 days but...441-B14-FRT£846.56 - £1,086.80 -

Shigella spp & EIEC RT-PCR (CE) | B12-FRT
£846.56 - £1,086.80Shigella spp & EIEC RT-PCR | B12-FRT from Sacace Biotechnologies is available for delivery Real Time PCR Kit for detection of Shigella spp andEnteroinvasive E.Coli (EIEC) Storage & Shipping : The kit can be shipped at 2-8°C for 3-4 days but...441-B12-FRT£846.56 - £1,086.80 -

Helicobacter pylori RT-PCR (CE) | B9-FR
£846.56 - £1,086.80Helicobacter pylori RT-PCR | B9-FR from Sacace Biotechnologies is available for delivery Real Time PCR kit for qualitative detection of Helicobacter pylori Storage & Shipping :The kit can be shipped at 2-8°C for 3-4 days but should be stored at 2-8°C and...441-B9-FR£846.56 - £1,086.80 -

Mycoplasma genitalium RT-PCR Quant (CE) | B4-FRT Q
£846.56 - £1,086.80Mycoplasma genitalium RT-PCR Quant (CE) | B4-FRT Q from Sacace Biotechnologies is available for delivery Real Time PCR Kit for quantitative detection of Mycoplasma genitalium Storage & Shipping : The kit can be shipped at 2-8°C for 3-4 days but...441-B4-FRT-Q£846.56 - £1,086.80 -

Ureaplasma species RT-PCR Quant | B2-FRT Q
£846.56 - £1,086.80Ureaplasma species RT-PCR Quant | B2-FRT Q from Sacace Biotechnologies is available for delivery Real Time PCR Kit for quantitative detection of Ureaplasma species Storage & Shipping : The kit can be shipped at 2-8°C for 3-4 days but should be...441-B2-FRT-Q£846.56 - £1,086.80 -

Parvovirus B19 RT-PCR Quant (CE) | V49-FRT
£846.56 - £1,741.17Parvovirus B19 RT-PCR Quant (CE) | V49-FRT from Sacace Biotechnologies is available for delivery Real Time PCR Kit for quantitative detection of Parvovirus B19 Storage & Shipping : The kit can be shipped at 2-8°C for 3-4 days but should be stored...441-V49-FRT£846.56 - £1,741.17 -

Swine Influenza Virus A/H1 RT-PCR (CE) | V55-FRT
£848.85 - £1,146.29Swine Influenza Virus A/H1 RT-PCR (CE) | V55-FRT from Sacace Biotechnologies is available for delivery Real-Time PCR test for the qualitative detection of Swine Influenza Virus A/H1 RNA Storage & Shipping : Transport Medium Plus should be transported...441-V55-FRT£848.85 - £1,146.29 -

Transport Medium for Swabs | R12-Resp
£848.85Transport Medium for Swabs | R12-Resp from Sacace Biotechnologies is available for delivery Reagent for transportation and storage of clinical material Storage & Shipping : Transport Medium Plus should be transported at 225 °C.. Transport Medium...441-R12-Resp£848.85 -

Transport Medium Plus | R12-Stab
£848.85Transport Medium Plus | R12-Stab from Sacace Biotechnologies is available for delivery Reagent for transportation and storage of clinical material Storage & Shipping : Transport Medium Plus should be transported at 225 °C.. Transport Medium Plus...441-R12-Stab£848.85 -

Cardio Thrombophilia Panel SNP | T01001-96-S
£848.85Cardio Thrombophilia Panel SNP | T01001-96-S from Sacace Biotechnologies is available for delivery Real Time PCR kit for detection of F2 20210 G>A , F5 1691 G>A (Arg506Gln), F7 10976 G>A (Arg353Gln), F13 G>T (Val34Leu), FGB -455 G>A, ITGA2...441-T01001-96-S£848.85 -

Folate Metabolism Panel SNP | T01002-96-S
£848.85Folate Metabolism Panel SNP (CE) | T01002-96-S from Sacace Biotechnologies is available for delivery Real Time PCR kit for detection of MTHFR 677 C>T , MTHFR 1298 A>C, MTR 2756 A>G, MTRR 66 A>G Storage & Shipping : Stable for up to one...441-T01002-96-S£848.85 -

Obesity & Diabetes Screen | T01372-96-S
£848.85Obesity & Diabetes Screen | T01372-96-S from Sacace Biotechnologies is available for delivery Real Time PCR test for dection of 8 single nucleotide polymorphisms (SNPs): PPARG2, ADRB2, ADRB3, FABP2, LPL, INS and FTO genes Storage & Shipping...441-T01372-96-S£848.85 -

Chlamydia trachomatis/Ureaplasma/ M.genitalium RT-PCR | TB46-100FRT
£848.85Chlamydia trachomatis/Ureaplasma/ M.genitalium RT-PCR | TB46-100FRT from Sacace Biotechnologies is available for delivery Multiplex Real Time PCR kit for qualitative detection of Chlamydia trachomatis, Ureaplasma species and Mycoplasma genitalium Storage...441-TB46-100FRT£848.85 -

Chlamydia trachomatis/Ureaplasma/ M.genitalium/M.hominis RT-PCR | TB60-100FRT
£848.85Chlamydia trachomatis/Ureaplasma/ M.genitalium/M.hominis RT-PCR | TB60-100FRT from Sacace Biotechnologies is available for delivery Multiplex Real Time PCR kit for qualitative detection of Chlamydia trachomatis, Ureaplasma species, Mycoplasma genitalium...441-TB60-100FRT£848.85 -

N.gonorrhoeae/C.trachomatis/ M.genitalium/T.vaginalis RT-PCR | TB61-100FRT
£848.85N.gonorrhoeae/C.trachomatis/ M.genitalium/T.vaginalis RT-PCR | TB61-100FRT from Sacace Biotechnologies is available for delivery Multiplex Real Time PCR kit for the detection of Neisseria gonorrhoeae, Chlamydia trachomatis, Mycoplasma genitalium and...441-TB61-100FRT£848.85 -

N.gonorrhoeae/C.trachomatis/ M.genitalium RT-PCR | TB67-100FRT
£848.85N.gonorrhoeae/C.trachomatis/ M.genitalium RT-PCR | TB67-100FRT from Sacace Biotechnologies is available for delivery Multiplex Real Time PCR kit for the detection of Neisseria gonorrhoeae, Chlamydia trachomatis and Mycoplasma genitalium Storage &...441-TB67-100FRT£848.85 -

T.vaginalis/N.gonorrhoeae/C.trachomatis RT-PCR | TB83-100FRT
£848.85T.vaginalis/N.gonorrhoeae/C.trachomatis RT-PCR | TB83-100FRT from Sacace Biotechnologies is available for delivery Multiplex Real Time PCR kit for qualitative detection of Chlamydia trachomatis, Neisseria gonorrhoeae and Trichomonas vaginalis Storage...441-TB83-100FRT£848.85 -

Toxoplasma gondii RT-PCR | TP1-50FRT
£848.85Toxoplasma gondii RT-PCR | TP1-50FRT from Sacace Biotechnologies is available for delivery Real Time PCR kit for qualitative detection of Toxoplasma gondii Storage & Shipping : Stable for up to one year when stored at 20°C, store it at 70°C...441-TP1-50FRT£848.85 -

CMV RT-PCR | TV7-100FR
£848.85CMV RT-PCR | TV7-100FR from Sacace Biotechnologies is available for delivery Real Time PCR kit for the qualitative detection of Cytomegalovirus (CMV) Storage & Shipping : Stable for up to one year when stored at 20°C, store it at 70°C or...441-TV7-100FR£848.85 -

Rubella RT-PCR Qual | TV24-50FRT
£848.85Rubella RT-PCR Qual | TV24-50FRT from Sacace Biotechnologies is available for delivery Real-Time PCR test for qualitative detection of Rubella Virus Storage & Shipping : Must be stored at 2-8°C.The kit can be shipped at 2-8°C but should be...441-TV24-50FRT£848.85 -

Bru-Com RT-PCR | TVET9-50FRT
£848.85Bru-Com RT-PCR | TVET9-50FRT from Sacace Biotechnologies is available for delivery Real Time Kit for use with Rotor-Gene 2000/3000/6000 (Corbett Research, Qiagen) Storage & Shipping : Stable for up to one year when stored at 20°C, store it at...441-TVET9-50FRT£848.85 -

Rubella RT-PCR Qual (CE) | V24-50FRT
£848.85Rubella RT-PCR Qual (CE) | V24-50FRT from Sacace Biotechnologies is available for delivery Real-Time PCR test for qualitative detection of Rubella Virus Storage & Shipping : Must be stored at 2-8°C.The kit can be shipped at 2-8°C but should...441-V24-50FRT£848.85 -

Chikungunya RT-PCR (CE) | V122-96FRT
£848.85Chikungunya RT-PCR | V122-96FRT from Sacace Biotechnologies is available for delivery Real Time PCR kit for qualitative detection of Chikungunya virus in clinical material Storage & Shipping : Must be stored at 2-8°C.The kit can be shipped at...441-V122-96FRT£848.85 -

Toxoplasma gondii RT-PCR (CE) | P1-50FRT
£848.85Toxoplasma gondii RT-PCR (CE) | P1-50FRT from Sacace Biotechnologies is available for delivery Real Time PCR kit for qualitative detection of Toxoplasma gondii Storage & Shipping : Must be stored at 2-8°C.The kit can be shipped at 2-8°C but...441-P1-50FRT£848.85 -

HLA-B27 RT-PCR | R116-50FRT
£848.85HLA-B27 RT-PCR | R116-50FRT from Sacace Biotechnologies is available for delivery Real Time PCR kit for detection of HLA-B27 polymorphism Storage & Shipping : Must be stored at 2-8°C.The kit can be shipped at 2-8°C but should be stored at...441-R116-50FRT£848.85 -

Express-DNA-A (CE) | K-2-17
£848.85Express-DNA-A | K-2-17 from Sacace Biotechnologies is available for delivery kit for storage of clinical materials and DNA purification Storage & Shipping : Must be stored at 2-8°C.The kit can be shipped at 2-8°C but should be stored at 2-8°C...441-K-2-17£848.85 -

DNA-Sorb-C (CE) | K-1-6/50
£848.85DNA-Sorb-C | K-1-6/50 from Sacace Biotechnologies is available for delivery Nucleic acid extraction kit for the extraction and purification of DNA from clinical materials Storage & Shipping : Must be stored at 2-8°C.The kit can be shipped at...441-K-1-6-50£848.85 -

Neisseria gonorrhoeae RT-PCR Quant (CE) | B204-100FRT
£848.85Neisseria gonorrhoeae RT-PCR Quant | B204-100FRT from Sacace Biotechnologies is available for delivery Real Time PCR Kit for quantitative detection of Neisseria gonorrhoeae Storage & Shipping : Must be stored at 2-8°C.The kit can be shipped at...441-B204-100FRT£848.85 -

Mycoplasma pneumoniae / Chlamydophila pneumoniae RT-PCR | TB42-4-FRT
£848.85 - £1,146.29Mycoplasma pneumoniae / Chlamydophila pneumoniae RT-PCR | TB42-4-FRT from Sacace Biotechnologies is available for delivery Real Time PCR kit for qualitative detection of Mycoplasma pneumoniae and Chlamydophila pneumoniae INTENDED USE : Kit Mycoplasma...441-TB42-4-FRT£848.85 - £1,146.29 -

Mycoplasma pneumoniae / Chlamydophila pneumoniae RT-PCR (CE) | B42-4-FRT
£848.85 - £1,146.29Mycoplasma pneumoniae / Chlamydophila pneumoniae RT-PCR (CE) | B42-4-FRT from Sacace Biotechnologies is available for delivery Real Time PCR kit for qualitative detection of Mycoplasma pneumoniae and Chlamydophila pneumoniae INTENDED USE : Kit...441-B42-4-FRT£848.85 - £1,146.29 -

Mycoplasma hominis RT-PCR Quant (CE) | B3-100FRT Q
£883.17Mycoplasma hominis RT-PCR Quant | B3-100FRT Q from Sacace Biotechnologies is available for delivery Background: Creative Biogene Mycoplasma Hominis (MH) Real Time PCR Kitis used for the detection of Mycoplasma Hominis (MH) in genital swabs or urine...441-B3-100FRT-Q£883.17 -

AZF System Y-chromosome (CE) | 01200-5
£883.17AZF System Y-chromosome | 01200-5 from Sacace Biotechnologies is available for delivery Real Time PCR test for detection of the microdeletions in AZF regions: AZFa (sY84, sY86), AZFb (sY127, sY134), AZFc (sY254, sY255) of the human Y chromosome. INTENDED...441-01200-5£883.17 -

West Nile Virus RT-PCR (Complete) (CE) | TV53-50FRT
£883.17West Nile Virus RT-PCR (Complete) | TV53-50FRT from Sacace Biotechnologies is available for delivery Complete Real Time PCR test with RNA purification kit INTENDED USE : WNV RT-PCR is Real-Time amplification test for the qualitative detection of West...441-TV53-50FRT£883.17 -

COVID-19 SARS-Cov-2 Variant Typing RT-PCR (CE) | V435-96FRT/V
£883.17COVID-19 SARS-Cov-2 Variant Typing RT-PCR | V435-96FRT/V from Sacace Biotechnologies is available for delivery Background: Real Time PCR kit for qualitative detection of SARS-CoV-2 Variant mutations in the Spike protein: HV 69-70 DEL, N501Y, E484K,...441-V435-96FRT-V£883.17 -

SARS-CoV-2/Influenza A/B multiplex RT-PCR (CE) | V440-100FRT
£883.17SARS-CoV-2/Influenza A/B multiplex RT-PCR | V440-100FRT from Sacace Biotechnologies is available for delivery Background: SARS-CoV-2/Influenza A/B multiplex RT-PCR is Real-Time PCR test for detection and differentiation of the SARS-CoV-2, Influenza A...441-V440-100FRT£883.17 -

SaMag-96 NEW (CE) | SM-12
£37,694.80SaMag-96 NEW | SM-12 from Sacace Biotechnologies is available for delivery Description: General information: CE IVD Automatic Nucleic Acids Extraction System (12 samples) Type: Instruments Storage and Shipping : stock441-SM-12-NEW£37,694.80 -

SaMag-12 (CE) | SM-12
£22,479.60SaMag-12 | SM-12 from Sacace Biotechnologies is available for delivery Description: General information: CE IVD Automatic Nucleic Acids Extraction System (12 samples) Type: Instruments Storage and Shipping : stock441-SM-12£22,479.60 -

SaCycler-96R 5x (CE) | SC-96R 5x
£27,124.24SaCycler-96R 5x | SC-96R 5x from Sacace Biotechnologies is available for delivery Description: General information: CE IVD Automatic Nucleic Acids Extraction System (12 samples) Type: Instruments Storage and Shipping : on request441-SC-96R 5x£27,124.24 -

SaCycler-96 IVD 5x (CE) | SC-96I 5x
£23,600.72SaCycler-96 IVD 5x | SC-96I 5x from Sacace Biotechnologies is available for delivery Description: General information: CE IVD 5 channels Real Time PCR 96-well format System (closed software version for Sacace Real Time PCR kits only) Type: Real Time...441-SC-96I 5x£23,600.72 -

M-Sorb-S NEW (CE) | K502/100/A
£518.23M-Sorb-S NEW | K502/100/A from Sacace Biotechnologies is available for delivery Description: General information: For the rapid, efficient magnetic preparation of highly pure viral nucleic acids from human nasopharyngeal swab specimens, sputum,...441-K502-100-A£518.23 -

SaMag Plant Extraction Kit (CE) | SM014
£441.58SaMag Plant Extraction Kit | SM014 from Sacace Biotechnologies is available for delivery Description: General information: For use with SaMag-12/24 instruments; extraction of DNA from Plant (leaf, seeds and spores) and fungal tissues. Target Disease...441-SM014£441.58 -

SaMag Viral/Bacterial Nucleic Acids Extraction Kit B (CE) | SM012
£507.94SaMag Viral/Bacterial Nucleic Acids Extraction Kit B | SM012 from Sacace Biotechnologies is available for delivery Description: General information: For use with SaMag-12/24 instruments; for extraction of viral and bacterial DNA/RNA from cell-rich...441-SM012£507.94 -

SaMagTotal RNA Extraction kit (CE) | SM015
£459.89SaMagTotal RNA Extraction kit | SM015 from Sacace Biotechnologies is available for delivery Description: General information: For use with SaMag-12/24 instruments; extraction of total RNA from clinical specimens Target Disease Type: RNA&DNA...441-SM015£459.89 -

SaMag FFPE Tissue DNA Extraction kit (CE) | SM009
£431.29SaMag FFPE Tissue DNA Extraction kit | SM009 from Sacace Biotechnologies is available for delivery Description: General information: For use with SaMag-12/24 instruments; extraction of DNA of from Formaldehyde Fixed-Paraffin Embedded tissue Target...441-SM009£431.29 -

SaMag Tissue DNA Extraction kit (CE) | SM004
£402.69SaMag Tissue DNA Extraction kit | SM004 from Sacace Biotechnologies is available for delivery Description: General information: For use with SaMag-12/24 instruments; extraction of DNA of from tissue Target Disease Type: RNA&DNA...441-SM004£402.69 -

SaMag Bacterial DNA Extraction kit (CE) | SM006
£332.90SaMag Bacterial DNA Extraction kit | SM006 from Sacace Biotechnologies is available for delivery Description: General information: For use with SaMag-12/24 instruments; extraction of bacterial DNA of from clinical specimens or coltures Target Disease...441-SM006£332.90 -

SaMag Blood DNA Extraction kit (CE) | SM001
£332.90SaMag Blood DNA Extraction kit | SM001 from Sacace Biotechnologies is available for delivery Description: General information: For use with SaMag-12/24 instruments; extraction of genomic DNA from whole blood, peripheral blood mononuclear cells or buffy...441-SM001£332.90 -

SaMag TB DNA Extraction kit (CE) | SM008
£402.69SaMag TB DNA Extraction kit | SM008 from Sacace Biotechnologies is available for delivery Description: General information: For use with SaMag-12/24 instruments; extraction of genomic DNA of Mycobacteria spp. (e.g. M.tuberculosis) from clinical...441-SM008£402.69 -

SaMag STD DNA Extraction kit (CE) | SM007
£321.46SaMag STD DNA Extraction kit | SM007 from Sacace Biotechnologies is available for delivery Description: General information: For use with SaMag-12/24 instruments; extraction of STD DNA (C.trachomatis, N.gonorrhoeae, HPV...) from swabs, urine sediment,...441-SM007£321.46 -

SaMag Viral Nucleic Acids Extraction kit (CE) | SM003
£321.46SaMag Viral Nucleic Acids Extraction kit | SM003 from Sacace Biotechnologies is available for delivery Description: General information: For use with SaMag-12/24 instruments; extraction of Viral nucleic acids from human biological specimens such as...441-SM003£321.46 -

Genomic column DNA Express (CE) | K-1-1/E
£345.49Genomic column DNA Express | K-1-1/E from Sacace Biotechnologies is available for delivery Description: General information: Genomic DNA from whole blood, plasma, buffy coat, body fluids, etc. Target Disease Type: RNA&DNA purification Specific...441-K-1-1-E£345.49 -

Ribo Virus Proteinase (CE) | K-2/C/P
£585.73Ribo Virus Proteinase | K-2/C/P from Sacace Biotechnologies is available for delivery Description: General information: For rapid purification of viral RNA/DNA (HCV,HBV,CMV...) from plasma or Target Disease Type: RNA&DNA purification Specific...441-K-2-C-P£585.73 -

Ribo Virus (CE) | K-2/C
£521.66Ribo Virus | K-2/C from Sacace Biotechnologies is available for delivery Description: General information: For rapid purification of viral RNA (HCV,HIV,HGV..) from plasma or serum Target Disease Type: RNA&DNA purification Specific Application: Spin...441-K-2-C£521.66 -

Magno-Virus/V (CE) | K-2-16/1000
£863.72Magno-Virus/V | K-2-16/1000 from Sacace Biotechnologies is available for delivery Description: General information: For rapid magnetic purification of viral RNA and DNA (e.g. HCV, HIV, HBV, HAV, HDV, Enteroviruses, CMV) from cell free body fluids such...441-K-2-16-1000£863.72 -

DNA/RNA-Prep NA (CE) | K-2-9/2
£393.54DNA/RNA-Prep NA | K-2-9/2 from Sacace Biotechnologies is available for delivery Description: General information: RNA/DNA purification kit from swabs, plasma, tissue, feces, etc Target Disease Type: RNA&DNA purification Specific Application: Silica...441-K-2-9-2£393.54 -

DNA/RNA-Prep (CE) | K-2-9
£345.49DNA/RNA-Prep | K-2-9 from Sacace Biotechnologies is available for delivery Description: General information: RNA/DNA purification kit from plasma, serum, liquor, tissue, feces, etc Target Disease Type: RNA&DNA purification Specific Application:...441-K-2-9£345.49 -

Rapid DNA (CE) | K-2-17
£236.81Rapid DNA | K-2-17 from Sacace Biotechnologies is available for delivery Description: General information: Fast DNA Extraction Kit from periodontal pockets, plaque and for genomic DNA Target Disease Type: RNA&DNA purification Specific Application:...441-K-2-17-1£236.81 -

Ribo-Sorb (CE) | K-2-1/100
£345.49Ribo-Sorb | K-2-1/100 from Sacace Biotechnologies is available for delivery Description: General information: RNA/DNA purification kit from plasma, serum, liquor, tissue, feces, etc Target Disease Type: RNA&DNA purification Specific Application:...441-K-2-1-100£345.49 -

DNA-Sorb-D (CE) | K-1/8-100
£417.56DNA-Sorb-D | K-1/8-100 from Sacace Biotechnologies is available for delivery Description: General information: DNA extraction kit from liquid-based cytology samples (Cytoscreen, PreservCyt,..) Target Disease Type: RNA&DNA purification Specific...441-K-1-8-100£417.56 -

DNA-Sorb-B (CE) | K-1-1/B/100
£297.44DNA-Sorb-B | K-1-1/B/100 from Sacace Biotechnologies is available for delivery Description: General information: DNA extraction kit from whole blood, plasma, liquor, sputum, bioptats, etc. Target Disease Type: RNA&DNA purification Specific...441-K-1-1-B-100£297.44
